Patient Lifting Equipment Market Report, Size and Forecast 2026–2033
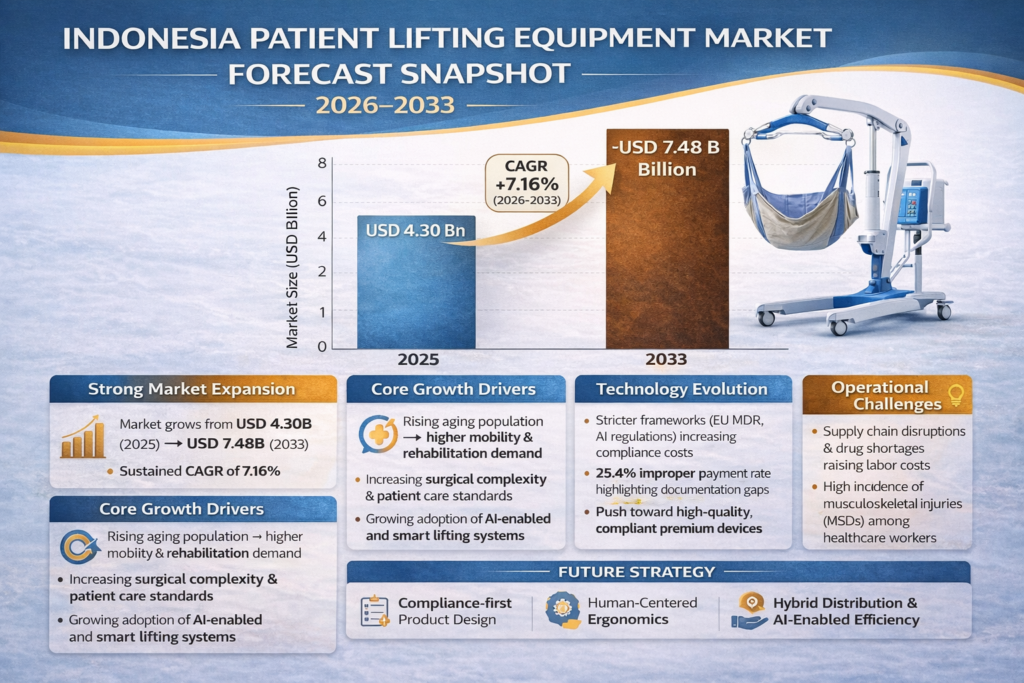
Patient Lifting Equipment Market Forecast Snapshot: 2026–2033
| Parameter | Value |
|---|---|
| Base Year | 2025 |
| Base Year Market Size | USD 4.30 Billion |
| Forecast Period | 2026–2033 |
| Forecasted CAGR | 7.16% |
| Forecasted Market Size (2033) | USD 7.48 Billion |
Market Size & Forecast
The Patient Lifting Equipment market is projected to expand from a base size of USD 4.30 billion in 2025 to approximately USD 7.48 billion by 2033, reflecting a compound annual growth rate (CAGR) of 7.16% over the forecast period. This growth trajectory reflects a confluence of several structural factors reshaping the healthcare delivery ecosystem and the attendant demand for patient handling solutions.
Fundamentally, this growth is driven by the increasing complexity of surgical procedures and patient care practices, which necessitate advanced lifting technologies to ensure safety, efficiency, and regulatory compliance. Hospitals and long-term care facilities are compelled to increase investment in specialized equipment to adapt to evolving clinical workflows. Notably, digital health applications and accelerated reimbursement pathways such as Germany’s DiGA program and France’s Transitional Coverage (PECT) scheme catalyze technology adoption, compressing the development-to-commercialization timeline for innovative lifting devices.
Market expansion is also influenced by demographic trends, including the aging global population, which elevates demand for mobility support and rehabilitative transfer assistance. However, the market’s growth is tempered by operational risks, including improper payment rates (notably a 25.4% improper payment incidence for patient lifts in 2024) and supply bottlenecks such as drug shortages that increase labor costs substantially (estimated at $360 million annually due to shortages’ impact on patient care).
Despite these challenges, the Patient Lifting Equipment market’s moderate competitive intensity and fragmented structure create opportunities for emerging technologies, particularly in smart assisted lifting systems and AI-driven platforms. These innovations enhance ergonomic safety and operational efficiency, further underpinning market growth. The CAGR of 7.16% encapsulates both the expanding demand and the ongoing capital investments required to meet increasingly stringent regulatory and clinical standards.
Market Overview
The Patient Lifting Equipment market is characterized by a fragmented competitive landscape with moderate intensity, where two tier-1 players coexist alongside numerous smaller providers and niche specialists. Major healthcare systems like Mayo Clinic and Kaiser Permanente exemplify early adopters of advanced patient handling technologies, including human-centered design (HCD) adaptations and active exoskeletons such as ALDAK, developed by GOGOA, which can assist caregivers in lifting weights up to 40 kg. These innovations address critical occupational health risks, particularly musculoskeletal disorders (MSDs), which remain disproportionately high among healthcare workers, with incidence rates for nursing assistants measured at 166.3 per 10,000 workers—more than five times the average for all industries.
The market’s operational model is predominantly hybrid, integrating both direct manufacturer sales and medical equipment distributors, supported by a growing online sales channel. Supply chain complexity is moderate but increasingly strained by drug shortages and medication supply disruptions, which indirectly impact patient handling through increased labor demands and operational costs. These bottlenecks compel healthcare providers to reassess procurement strategies, emphasizing reliability and patient-centered design in equipment selection.
Regulatory oversight is intensifying, driven by evolving frameworks such as the European Union’s Regulations (EU) 2017/745 and 2017/746 on medical devices and in vitro diagnostics, alongside the newly enacted Regulation (EU) 2024/1689 addressing artificial intelligence in medical contexts. These regulations elevate compliance requirements related to safety, efficacy, and post-market surveillance, thereby raising market entry and operational execution standards. The introduction of centralized information hubs like the European Database on Medical Devices (EUDAMED), expected by 2027, will further enhance transparency and traceability.
Investment trends display rising capital intensity, with healthcare facilities prioritizing projects that cover the weighted average cost of capital (WACC). Average Series A and C funding rounds in healthcare technology have increased, indicating maturation and investor confidence in innovative patient lifting solutions. Notably, large investments such as Target’s $550 million infusion into logistics infrastructure exemplify the capital scale necessary for scaling distribution and deployment capabilities.
Technologically, the market is in a growth maturity stage, characterized by high innovation intensity and moderate patent activity. Emerging technologies include AI-driven platforms that accelerate regulatory compliance and documentation, sensor-assisted lift systems with automated safety monitoring, and digital health applications that facilitate reimbursement and adoption. The precision and recall performance of institutional disambiguation solutions (0.96 and 0.95 respectively) underscore the increasing application of AI in streamlining healthcare operations.
Altogether, the Patient Lifting Equipment market is shaped by intersecting drivers: the imperative for ergonomic safety amid high occupational injury rates; evolving regulatory frameworks that reprice risk and compliance; capital deployment accelerating capacity scaling; and competitive dynamics influenced by value-chain reconfiguration and technology commercialization.
Structural Drivers of Market Growth
The growth of the Patient Lifting Equipment market is driven by four interrelated structural factors synthesized from cross-dimensional signals that collectively explain why and how the market is evolving.
1. Innovation and Commercialization Acceleration
The increasing maturity of technology and availability of targeted funding are compressing the time from development to commercialization of patient lifting equipment, expanding the addressable applications and accelerating adoption rates. Surgical operations are becoming more specialized and complex, necessitating advanced lifting devices to support both patients and caregivers safely. Healthcare organizations are compelled to allocate greater investment capital to procure these technologies, as hospitals increasingly prioritize equipment that integrates digital health functionalities and AI-driven compliance tools.
Programs like Germany’s DiGA and France’s PECT facilitate faster market entry and provisional reimbursement, reducing financial barriers and encouraging manufacturers to innovate. These schemes provide up to 12 months of temporary reimbursement while products await permanent listing, thus supporting early adoption and market penetration. This regulatory facilitation dovetails with rising investor interest, as demonstrated by average Series A funding of $18.7 million and Series C rounds averaging $50 million, which supply critical capital for development and scaling.
The implication is a market that is rapidly diversifying its product portfolio—from traditional manual and electric lifts to smart, sensor-assisted systems equipped with automated safety monitoring. This innovation trajectory not only broadens clinical use cases but also helps alleviate caregiver injury risks by improving ergonomics and reducing physical strain, thereby aligning clinical needs with regulatory expectations and investment dynamics.
2. Compliance and Risk Repricing
Heightened regulatory scrutiny and emergent risk signals are reshaping operating requirements, leading to a repricing of compliance costs and elevated execution standards throughout the patient lifting equipment market. The implementation of stringent EU medical device regulations (2017/745, 2017/746) and the AI-specific Regulation (EU) 2024/1689 impose more rigorous safety and documentation demands, compelling manufacturers to invest in comprehensive post-market surveillance and quality assurance processes.
Concurrently, risk factors such as the high improper payment rate for patient lifts, measured at 25.4% in 2024, and the predominance of insufficient documentation (accounting for 91.8% of improper payments) spotlight operational vulnerabilities that increase financial exposure and compliance burdens for providers. Additionally, musculoskeletal disorder incidence among healthcare workers remains alarmingly high, with direct and indirect back injury costs estimated at $20 billion annually. These human and financial risks incentivize greater adoption of mechanically assisted patient lifts and novel ergonomic designs that reduce caregiver strain.
Insurance reimbursement frameworks and coverage policies also recalibrate product roadmaps, as evidenced by the temporary reimbursement mechanisms under PECT and DiGA. Providers’ inability to deliver preferred treatments due to drug shortages (reported by 71% of providers in 2017) further compounds risk by straining care delivery workflows, underscoring the necessity of reliable, compliant patient handling solutions.
As a result, market participants are compelled to enhance product robustness, documentation rigor, and regulatory alignment, which raises barriers to entry but simultaneously creates a premium segment for compliant, risk-mitigated devices that can command higher margins and foster trust among healthcare purchasers.
3. Competitive and Value-Chain Reconfiguration
The Patient Lifting Equipment market is undergoing a reallocation of bargaining power and competitive positioning due to shifting organizational, cultural, and supply chain dynamics. The fragmented market structure, with only two tier-1 players, reflects both moderate competitive intensity and significant room for smaller innovators, especially those addressing unmet ergonomic and safety needs.
Operational constraints within healthcare value chains—such as drug shortages causing an estimated $360 million per year in additional labor costs—exert pressure on procurement and deployment strategies. Approximately 70% of providers were able to pass on cost increases in 2024; however, many failed to meet growth targets, signaling margin compression and competitive challenges.
Organizational and cultural factors influence adoption rates of mechanical lifting equipment, with institutions like Mayo Clinic and Kaiser Permanente pioneering adaptations of human-centered design (HCD) and advanced assistive devices such as GOGOA’s ALDAK exoskeleton. These devices, which can lift up to 40 kg, represent a competitive differentiation aligned with caregiver safety and operational efficiency.
This reconfiguration forces portfolio repositioning, with companies emphasizing integration of patient-centered design, hybrid distribution models, and technology-enabled service offerings. The hybrid operational and distribution structure reflects the need to balance direct manufacturer relationships with institutional procurement and e-commerce channels, optimizing reach and responsiveness.
Consequently, competitive advantages increasingly derive from the ability to navigate value-chain complexities, leverage cultural buy-in, and manage cost pressures while delivering differentiated technologies that align with evolving clinical and regulatory demands.
4. Capital and Capacity Scaling
Substantial capital deployment is fueling capacity expansion and process upgrades across the Patient Lifting Equipment market, enabling faster scale-up in high-demand segments such as acute care hospitals and long-term care facilities. Investments by healthcare providers are driven by the imperative to manage increasingly complex surgical and patient care scenarios, which require specialized equipment and enhanced throughput.
The integration of reimbursement schemes like PECT, which offers up to 12 months of temporary coverage, reduces deployment friction by mitigating payor risk during the commercialization phase. This financial de-risking encourages hospitals to invest confidently in advanced lifting solutions.
Moreover, healthcare systems allocate approximately 10% of GDP to healthcare spending, underscoring the scale of capital available for upgrading infrastructure and technology. Large-scale investments, exemplified by Target’s $550 million funding in logistics infrastructure, also indicate the broader ecosystem’s readiness to support supply chain modernization, which is critical for timely distribution and installation of patient lifting devices.
Technological innovations such as AI-driven documentation, sensor-assisted lifts, and active exoskeletons require specialized manufacturing and service capabilities, necessitating capital-intensive capacity building. This scaling enhances market accessibility, reduces lead times, and improves after-sales support, collectively driving accelerated adoption.
Ultimately, capital and capacity scaling not only improve operational efficiency for providers but also increase market liquidity, fostering innovation diffusion and amplifying the Patient Lifting Equipment market’s growth potential during the forecast period.
Market Segmentation Analysis
Top-Level Segment Share Split
By End User: 30.00% (End users such as hospitals and healthcare facilities are primary buyers.)
By Technology: 25.00% (Technological advancements drive market growth.)
By Application: 15.00% (Different applications in patient care influence market segmentation.)
By Product Type: 20.00% (Various product types cater to specific needs in patient lifting.)
By Distribution Channel: 10.00% (Distribution channels impact market reach and accessibility.)
The Patient Lifting Equipment market exhibits substantial heterogeneity across multiple segmentation dimensions: end user, technology, application, product type, and distribution channel. These segments collectively define the market’s structural composition and inform targeted strategic approaches.
Regional Market Dynamics
Competitive Landscape
The competitive environment of the Patient Lifting Equipment market is marked by moderate intensity within a fragmented market structure, comprising numerous small and medium enterprises alongside a limited number of tier 1 players. The presence of only two tier 1 players underscores a landscape where dominant entities coexist with a broad base of niche and regional manufacturers.
Leading healthcare providers such as Mayo Clinic and Kaiser Permanente serve as influential stakeholders rather than direct equipment manufacturers, yet their strategic adoption and adaptation of advanced human-centered design (HCD) principles reinforce demand for innovative patient lifting solutions. Both institutions have integrated HCD methodologies, leveraging interdisciplinary inputs from anthropology, sociology, and human-computer interaction to optimize device usability and caregiver safety. Their influence extends indirectly through pilot programs and validation studies that shape market standards and accelerate clinical adoption of new technologies.
Among manufacturers, GOGOA distinguishes itself through the development of ALDAK, an active exoskeleton capable of assisting workers in lifting loads up to 40 kg. This capability not only addresses ergonomic challenges but also exemplifies the intersection of assistive technology and robotics within the market’s innovation frontier. The technological sophistication embedded in ALDAK positions GOGOA as a leader in addressing both labor efficiency and injury risk reduction, critical competitive advantages in a sector where back injuries cost healthcare systems billions annually.
Market entry barriers remain moderate due to the fragmented nature and relatively low capital intensity required for certain manual and electric lifting systems. However, scaling challenges arise from stringent regulatory compliance, complex value chains, and the need for integration with healthcare providers’ workflows. Larger players sustain their market positions by leveraging capabilities in technology innovation, regulatory navigation, and established distribution networks. For instance, companies embedding AI-driven platforms to streamline regulatory updates and device documentation reduce time-to-market and mitigate compliance risks, conferring competitive agility.
The market also reflects a dynamic interplay between competitive moves and value-chain constraints. Organizational and cultural factors within hospitals critically influence adoption rates of mechanical lift equipment, often determining safety outcomes more than device features alone. This creates opportunities for players who can tailor solutions to specific institutional contexts and provide comprehensive training and support services. The moderate competitive intensity level further suggests that while competition is robust, it is not overly aggressive, allowing firms to cultivate long-term client relationships and focus on incremental innovation.
Moreover, the limited number of tier 1 players indicates potential for consolidation or strategic partnerships, especially as investment capital rises and technology converges with digital health applications. The absence of recent significant M&A activity reflects either ongoing evaluation of market valuations or a strategic preference for organic growth amidst regulatory tightening and risk repricing pressures.
In summary, the Patient Lifting Equipment market’s competitive landscape is shaped by a balance of innovation-driven differentiation, regulatory complexity, and organizational adoption barriers, all underscored by a fragmented supply base where a few key players establish leadership grounded in technological and clinical expertise.
Strategic Outlook
The strategic horizon for the Patient Lifting Equipment market is defined by the convergence of innovation acceleration, regulatory compliance demands, and evolving value-chain configurations, all set against a backdrop of increasing healthcare complexity and demographic imperatives. Forward-looking market participants must navigate these intertwined forces to capture growth and sustain competitive advantage.
Innovation and commercialisation acceleration represent a core strategic imperative. The maturation of assistive technologies—such as AI-driven safety monitoring lifts and active exoskeletons—facilitates compression of development-to-commercialization timelines. Programs like Germany’s DiGA and France’s PECT establish precedents for provisional reimbursement schemes that reduce market entry delays and financial risks for novel devices. Firms investing in R&D aligned with such regulatory incentives can rapidly expand addressable applications beyond traditional hospital settings into home care and rehabilitation, thereby broadening revenue streams. Moreover, integrating digital health capabilities enhances device functionality and patient-centric outcomes, increasingly demanded by healthcare providers.
Simultaneously, compliance and risk repricing exert strategic pressure to elevate operating standards. The tightening of regulations, exemplified by Regulation (EU) 2024/1689 on artificial intelligence and the enforcement of medical device regulations (EU 2017/745 and 2017/746), compels manufacturers to embed robust quality management and post-market surveillance systems. Failure to comply not only risks market exclusion but also amplifies improper payment rates—as evidenced by the 25.4% improper payment rate for patient lifts driven largely by insufficient documentation (91.8%). Consequently, firms adopting sophisticated regulatory intelligence platforms and investing in compliance-centric organizational cultures will mitigate risk exposure and enhance market credibility.
Competitive and value-chain reconfiguration demands strategic agility. The influence of organizational culture on equipment adoption, coupled with the substantial labor costs linked to drug and equipment shortages, necessitates a reallocation of bargaining power along the value chain. Manufacturers and suppliers must increasingly partner with healthcare institutions to co-develop solutions tailored to workflow realities, thereby improving safety outcomes and cost-effectiveness. Strategic alliances with logistics providers and distributors can also optimize supply chain responsiveness, addressing bottlenecks that currently inflate operational costs by up to 10%.
Capital and capacity scaling remain essential for meeting burgeoning demand, particularly as surgical procedures grow more complex and hospitals seek to upgrade processes. High capital intensity requires judicious deployment of funds into manufacturing automation, supply chain digitization, and workforce training. The rising trend in Series A to C funding rounds, averaging from $18.7 million to $50 million, signals investor confidence in scaling technologically advanced patient lifting solutions. Companies that effectively leverage this capital influx to enhance throughput and reduce deployment friction will secure market leadership. Additionally, alignment with healthcare providers’ cost of capital (WACC) expectations ensures project viability and facilitates broader adoption.
Furthermore, strategic differentiation will increasingly hinge on hybrid product portfolios that blend manual, electric, and smart assisted lifting systems. Tailoring offerings across end users—from acute care hospitals to elderly home care settings—and diversifying distribution channels, including direct-to-consumer e-commerce platforms, will optimize market reach. Emphasizing patient-centered design and safety outcomes not only meets regulatory and reimbursement criteria but also aligns with healthcare providers’ ethical mandates, as exemplified by the British NHS’s shared decision-making principle: “no decision about me, without me.”
In essence, the strategic outlook for the Patient Lifting Equipment market is one of integrated innovation, compliance rigor, collaborative value-chain management, and capital-efficient scaling, all orchestrated to meet the shifting demands of modern healthcare delivery.
Final Market Perspective
The Patient Lifting Equipment market’s trajectory toward USD 7.48 billion by 2033 at a CAGR of 7.16% encapsulates the interplay of demographic imperatives, technological innovation, regulatory evolution, and value-chain reconfiguration. This landscape demands that market participants not only innovate but also adeptly manage risk and compliance while fostering collaborative relationships across complex supply chains.
Emerging reimbursement models and accelerated regulatory pathways fundamentally alter commercialization dynamics, enabling faster introduction of advanced lifting technologies that address both patient safety and caregiver ergonomics. However, the rising standards of regulatory compliance coupled with high improper payment risks mandate investments in documentation integrity and post-market surveillance, reshaping operational priorities and cost structures.
The competitive environment, characterized by moderate intensity and fragmentation, reflects a nuanced balance where established players leverage technological and clinical expertise to sustain leadership, while smaller innovators contribute through targeted solutions addressing niche requirements. The influence of organizational culture on adoption underscores the importance of contextualized engagement strategies and training programs that align technology with end-user realities.
Capital deployment into capacity and process upgrades emerges as a critical enabler, with investor confidence reflected in substantial funding rounds and logistics investments. The capacity to scale efficiently while adhering to cost of capital benchmarks will determine which players consolidate market share in high-demand segments.
Finally, regional disparities in regulation, healthcare infrastructure, and demographic pressures necessitate tailored approaches that respect local market conditions while capitalizing on global innovation and compliance trends. The hybrid operational and distribution models illustrate the sector’s adaptability but also highlight the ongoing need to resolve supply chain bottlenecks, including those caused by drug shortages.
In sum, the Patient Lifting Equipment market’s future is shaped by an intricate synthesis of innovation, compliance, competitive positioning, and capital efficiency, all oriented toward safer, more effective patient handling solutions that respond to the evolving demands of healthcare systems worldwide.
Research Methodology
This study employs a comprehensive mixed-methods approach, integrating both primary and secondary research to ensure a robust and multidimensional analysis of the Patient Lifting Equipment market in 2025. Primary research was conducted through in-depth interviews with a strategically selected panel of industry stakeholders, including manufacturers and distributors of patient lifting devices, procurement leaders from hospitals and clinics, frontline clinicians, regulatory and quality assurance specialists, as well as reimbursement and payer representatives where applicable. These expert insights provided critical qualitative and quantitative data, enabling a nuanced understanding of market dynamics, technology adoption trends, and end-user requirements across diverse healthcare settings such as acute care hospitals, intensive care units, home care environments, and long-term care facilities.
Secondary research was undertaken via a sophisticated triangulation methodology, drawing from a curated collection of authoritative databases and regulatory repositories. Sources encompassed peer-reviewed medical and scientific literature (PMC, PubMed), government and regulatory frameworks (Federal Register, OSHA, CMS, EMA, HSE UK), industry compliance and reimbursement guidelines, and economic and legal analyses from reputable platforms. This multi-source verification allowed for cross-validation of market size estimates, technology efficacy, regulatory impact, and reimbursement landscapes, ensuring the elimination of data bias and enhancing the report’s credibility. The triangulated data synthesis provided a solid foundation for trend extrapolation and competitive benchmarking, underpinning the strategic insights presented throughout the report. Collectively, this rigorous research design facilitates a comprehensive and actionable market intelligence resource for stakeholders engaged in the evolving Patient Lifting Equipment sector.
Table of Contents
Executive Summary
1.1 Market Forecast Snapshot (2026–2033)
1.2 Global Market Size & CAGR Analysis
1.3 Largest & Fastest-Growing Segments
1.4 Region-Level Leadership & Growth Trends
1.5 Key Market Drivers
1.6 Competitive Landscape Overview
1.7 Strategic Outlook Through 2033
Introduction & Market Overview
1.1 Definition of the Patient Lifting Equipment Market
1.2 Scope of the Study
1.3 Industry Evolution & Market Development
1.4 Supply Chain & Distribution Infrastructure
1.5 Impact of Consumer Trends
1.6 Regulatory & Compliance Landscape
1.7 Technology & Innovation Landscape
Research Methodology
1.1 Primary Research
1.2 Secondary Research
1.3 Market Size Estimation Model
1.4 Forecast Assumptions (2026–2033)
1.5 Data Validation & Triangulation
Market Dynamics
1.1 Drivers
1.1.1 Aging Population & Rising Elderly Care Demand
1.1.2 Increasing Healthcare Infrastructure Expansion
1.1.3 Growing Focus on Patient Safety & Caregiver Efficiency
1.1.4 Technological Advancements in Lifting Systems
1.1.5 Rising Demand for Home Healthcare Solutions
1.2 Restraints
1.2.1 High Equipment Costs
1.2.2 Limited Awareness in Emerging Markets
1.2.3 Infrastructure Constraints in Low-Income Regions
1.2.4 Maintenance & Operational Challenges
1.3 Opportunities
1.3.1 Expansion of Home Healthcare Market
1.3.2 Smart & Sensor-Based Lifting Systems
1.3.3 Emerging Market Penetration
1.3.4 Product Innovation & Customization
1.4 Challenges
1.4.1 Supply Chain Complexity
1.4.2 Regulatory Compliance & Certification
1.4.3 Market Fragmentation
1.4.4 Competitive Pricing Pressure
Patient Lifting Equipment Market Analysis (USD Billion), 2026–2033
1.1 Market Size Overview
1.2 CAGR Analysis
1.3 Regional Revenue Distribution
1.4 Segment Revenue Analysis
1.5 Distribution Channel Analysis
1.6 End-User Impact Analysis
Market Segmentation (USD Billion), 2026–2033
1.1 By End User
1.1.1 Hospitals
1.1.1.1 Acute Care Hospitals
1.1.1.1.1 Intensive Care Units
1.1.2 Home Care Settings
1.1.2.1 Elderly Home Care
1.1.2.1.1 Home Healthcare Services
1.1.3 Long-Term Care Facilities
1.1.3.1 Nursing Homes
1.1.3.1.1 Assisted Living Facilities
1.2 By Technology
1.2.1 Manual Lifting Systems
1.2.1.1 Hydraulic Lifting Mechanisms
1.2.1.1.1 Manual Patient Transfer Devices
1.2.2 Electric Lifting Systems
1.2.2.1 Motorized Lifting Mechanisms
1.2.2.1.1 Battery Powered Patient Transfer Devices
1.2.3 Smart Assisted Lifting Systems
1.2.3.1 Sensor-Assisted Lift Systems
1.2.3.1.1 Automated Safety Monitoring Lifts
1.3 By Application
1.3.1 Elderly Care
1.3.1.1 Assisted Living Patient Mobility
1.3.1.1.1 Long-Term Care Transfer Support
1.3.2 Patient Transfer
1.3.2.1 Bed to Wheelchair Transfer
1.3.2.1.1 Assisted Mobility Transfer
1.3.3 Rehabilitation Assistance
1.3.3.1 Mobility Rehabilitation Support
1.3.3.1.1 Physical Therapy Transfer Assistance
1.4 By Product Type
1.4.1 Ceiling Lifts
1.4.1.1 Fixed Ceiling Lifts
1.4.1.1.1 Track-Based Ceiling Lifts
1.4.1.1.1.1 Overhead Patient Transfer Systems
1.4.2 Sit-to-Stand Lifts
1.4.2.1 Powered Sit-to-Stand Lifts
1.4.2.1.1 Assisted Standing Devices
1.4.2.1.1.1 Rehabilitation Standing Lifts
1.4.3 Electric Floor Lifts
1.4.3.1 Battery Powered Patient Lifts
1.4.3.1.1 Mobile Electric Patient Lifts
1.4.3.1.1.1 Powered Transfer Lifts
1.4.4 Floor-Based Patient Lifts
1.4.4.1 Manual Floor Lifts
1.4.4.1.1 Hydraulic Floor Lifts
1.4.4.1.1.1 Portable Patient Floor Lifts
1.5 By Distribution Channel
1.5.1 Online Sales Channels
1.5.1.1 E-Commerce Medical Platforms
1.5.1.1.1 Direct-to-Consumer Mobility Equipment
1.5.2 Direct Manufacturer Sales
1.5.2.1 Healthcare Facility Contracts
1.5.2.1.1 Long-Term Care Facility Sales
1.5.3 Medical Equipment Distributors
1.5.3.1 Patient Handling Equipment Suppliers
1.5.3.1.1 Institutional Procurement
Market Segmentation by Geography
1.1 North America
1.2 Europe
1.3 Asia-Pacific
1.4 Latin America
1.5 Middle East & Africa
Competitive Landscape
1.1 Market Share Analysis
1.2 Product Portfolio Benchmarking
1.3 Product Positioning Mapping
1.4 Distribution & Supply Chain Partnerships
1.5 Competitive Intensity & Differentiation
Company Profiles
Strategic Intelligence & Pheonix AI Insights
1.1 Pheonix Demand Forecast Engine
1.2 Supply Chain & Infrastructure Analyzer
1.3 Technology & Innovation Tracker
1.4 Product Development Insights
1.5 Automated Porter’s Five Forces Analysis
Future Outlook & Strategic Recommendations
1.1 Expansion of Home Healthcare Solutions
1.2 Smart Lifting Technology Adoption
1.3 Product Innovation & Cost Optimization
1.4 Regional Expansion Strategies
1.5 Long-Term Market Outlook (2033+)
Appendix
About Pheonix Research
Disclaimer
Competitive Landscape
Patient Lifting Equipment Competitive Intensity & Market Structure Overview
The patient lifting equipment market is characterized by a fragmented structure, with a moderate level of competitive intensity. This structure is primarily driven by the presence of numerous small and medium-sized enterprises, alongside a few dominant players that significantly influence market dynamics. The fragmentation in the market allows for a diverse range of products and services, enhancing the options available to healthcare providers. However, it also poses challenges in terms of standardization and market penetration.
Two Tier 1 players, Mayo Clinic and Kaiser Permanente, stand out due to their strategic positioning and significant influence within the healthcare sector. These entities leverage their extensive networks and resources to drive innovation and set benchmarks for other players. Their involvement in this market is indicative of a broader trend where established healthcare providers are increasingly focusing on integrating advanced patient lifting solutions to improve patient care and operational efficiency.
The competitive landscape is further enriched by companies like GOGOA Mobility Robots, which have introduced innovative solutions such as the ALDAK active exoskeleton. This development underscores a shift towards more technologically advanced equipment that not only enhances patient safety but also aids healthcare professionals in performing their tasks more efficiently.
Patient Lifting Equipment Competitive Intensity & Market Structure Current Scenario
Leading Company Profiles
Mayo Clinic: Healthcare Provider. Leader in patient care and technology adoption.
Kaiser Permanente: Healthcare Provider. Integrated healthcare system with focus on innovation.
GOGOA Mobility Robots: Assistive Technology Developer. Developer of advanced mobility solutions.
Strong Arm: Patient Lifting Equipment Manufacturer. Specializes in ergonomic lifting solutions.
NIOSH: Research and Standards Organization. Sets safety standards for patient lifting equipment.
Participatory co-design: Design Consultancy. Focuses on user-centered design for healthcare solutions.
Electronic Health Records: Technology Provider. Integrates patient lifting data into healthcare systems.
Human-centered design: Design Approach. Enhances usability and safety of patient lifting equipment.
Key Competitive Intensity & Market Structure Signals in Patient Lifting Equipment
In the patient lifting equipment market, several key signals highlight the dynamics of competitive intensity and market structure. Primarily, the adoption and implementation of mechanical patient lift equipment are heavily influenced by organizational and cultural factors. These aspects are critical in enhancing safety outcomes, as they dictate how well institutions can integrate new technologies into their existing systems. For example, larger healthcare systems like Mayo Clinic and Kaiser Permanente have been able to adapt human-centered design (HCD) approaches effectively. By focusing on innovation and technology adoption, these organizations set benchmarks in patient safety and equipment usability, thus maintaining a strategic advantage over smaller competitors.
Another significant signal is the low barriers to entry in the market. This trait encourages a broad spectrum of providers, including small and rural practices, to participate actively. While this influx increases competition and product variety, it also leads to challenges in standardization and quality control. As smaller entities enter the market, they are often able to offer competitive pricing, which can lead to more affordable options for healthcare providers. However, the lack of significant capital and infrastructure often hampers their ability to scale operations or invest in advanced technologies.
The economic environment also plays a crucial role. For instance, 70 percent of healthcare providers successfully passed on cost increases in 2024, yet many failed to reach growth targets. This indicates a challenging landscape where companies must balance cost management with the need for growth and innovation. The ability to pass costs on successfully demonstrates strong pricing power but also highlights the potential limits of market expansion when growth targets are unmet.
The aftermath of the COVID-19 crisis further underscores the importance of scale and resource consolidation. Multihospital systems have demonstrated superior project management capabilities compared to smaller hospitals, which struggled more significantly. This scenario highlights the strategic advantage of larger players in terms of resilience and operational efficiency, allowing them to maintain a robust market presence despite economic disruptions.
Strategic Implications of Competitive Intensity & Market Structure in Patient Lifting Equipment
The implications of these competitive intensity signals are multifaceted, shaping both strategic decisions and market outcomes. For companies operating within this fragmented market, differentiation becomes a critical strategy. The development and introduction of innovative products, such as GOGOA’s ALDAK exoskeleton, are vital for gaining a competitive edge. By offering advanced solutions that enhance patient safety and operational efficiency, companies can strengthen their market position and appeal to a broader customer base.
Nevertheless, the low barriers to entry mean that new entrants can quickly emerge, potentially saturating the market with a variety of alternatives. This situation compels established players to continuously innovate and adapt to changing demands. Companies like Strong Arm, specializing in ergonomic lifting solutions, must focus on developing unique value propositions and leveraging technological advancements to stay ahead.
Cost management remains a critical challenge. The fact that a significant percentage of healthcare providers managed to pass on cost increases yet struggled with growth highlights the need for strategic pricing and cost control. Companies must find ways to optimize their operations and streamline processes to maintain profitability without compromising on quality or innovation.
Larger organizations, such as Mayo Clinic and Kaiser Permanente, are well-positioned to capitalize on these dynamics due to their scale and resources. Their ability to integrate cutting-edge technology and maintain a focus on patient-centered care allows them to set industry standards and influence market trends. Smaller players must develop niche strategies, focusing on specific market segments or geographic areas where they can establish a strong foothold.
Patient Lifting Equipment Competitive Intensity & Market Structure Forward Outlook
Looking ahead, the patient lifting equipment market is poised to experience continued evolution driven by technological advancements and changing healthcare demands. The integration of digital technologies and the emphasis on enhancing patient outcomes will likely remain central to strategic initiatives. Companies that can effectively combine innovation with operational efficiency are expected to thrive in this competitive environment.
The market structure is anticipated to remain fragmented, but with potential consolidation as larger players seek to expand their influence and capabilities. Acquisitions and partnerships may become more prevalent as companies aim to access new technologies or enter untapped markets. For instance, collaborations between technology providers and healthcare institutions could lead to innovative solutions that address specific needs.
The role of regulatory frameworks and reimbursement schemes will also be crucial. Policies that support the adoption of advanced healthcare technologies can encourage investment and innovation. However, companies must navigate these regulations carefully to ensure compliance and maximize opportunities.
In conclusion, the patient lifting equipment market will continue to be shaped by a complex interplay of factors including organizational dynamics, economic conditions, and technological innovation. Companies that can strategically adapt to these changes by leveraging their strengths and addressing market challenges will be well-positioned for sustained success. The emphasis on safety, efficiency, and patient-centered care will be pivotal in defining competitive strategies and market leadership in the years to come.
Value Chain
Patient Lifting Equipment Value Chain & Supply Chain Evolution Overview
In the healthcare industry, the value chain and supply chain dynamics of patient lifting equipment serve as critical determinants of market structure, influencing everything from pricing strategies to competitive behavior. The evolution of supply chains in this sector is intricately tied to operational models, regulatory compliance, and technological innovations that aim to enhance patient care and safety. The primary operational model for patient lifting equipment is hybrid, integrating both traditional and modern supply chain methodologies. This model accommodates a distribution structure that is also hybrid, facilitating both direct and indirect channels to ensure that equipment reaches end-users efficiently.
The complexity level of the supply chain for patient lifting equipment is moderate, reflecting a balance between the need for specialized components and the ability to adapt to technological advancements. Key stages within this value chain include the evaluation of projects, coverage of innovative technology, and patient-centered design. Each stage necessitates a careful orchestration of resources and capabilities, underscoring the importance of strategic alignment across the chain to mitigate bottlenecks and capitalize on emerging opportunities.
In this context, drug shortages and the availability of medications are notable bottlenecks that can impact the supply chain’s efficiency. These shortages not only affect the timely delivery of patient lifting equipment but also have a ripple effect on healthcare operations, potentially increasing operational costs and affecting patient outcomes. Effective supply chain management can mitigate these challenges by optimizing inventory tracking to identify potential shortages and streamline logistics.
The European Database on Medical Devices (EUDAMED), expected to become mandatory around 2027, will serve as a centralized hub for information on medical devices, including patient lifting equipment. This regulatory development underscores the growing importance of compliance and data transparency in the value chain, influencing how companies design, manufacture, and distribute their products. Furthermore, the Medicare Coverage of Innovative Technology (MCIT) program highlights the critical role of regulatory frameworks in defining what is considered ‘reasonable and necessary’ for medical devices, impacting the adoption of innovative patient lifting solutions.
Patient Lifting Equipment Value Chain & Supply Chain Evolution Current Scenario
Market-Specific Value Chain
- Research and Development: Innovation and design of new patient lifting solutions.
- Manufacturing: Production of patient lifting equipment.
- Regulatory Compliance: Ensuring equipment meets safety and regulatory standards.
- Distribution: Logistics and supply chain management for equipment delivery.
- End User Implementation: Deployment and use of equipment in healthcare settings.
Company-to-Stage Mapping
- Research and Development: No mapped companies
- Manufacturing: Strong Arm, GOGOA Mobility Robots
- Regulatory Compliance: NIOSH, Participatory co-design, Human-centered design
- Distribution: Electronic Health Records
- End User Implementation: Mayo Clinic, Kaiser Permanente
Key Value Chain & Supply Chain Evolution Signals in Patient Lifting Equipment
The evolution of the value chain and supply chain in the patient lifting equipment market is driven by several key signals that shed light on current dynamics and future trajectories. One of the most pressing signals is the impact of drug shortages on healthcare operations and patient outcomes. These shortages not only increase operational costs but also affect the ability of healthcare providers to deliver timely and effective care, underscoring the importance of effective supply chain management to mitigate these challenges.
Supply chain management plays a vital role in ensuring that hospitals can track inventory to quickly identify possible shortages or any other issues with product availability. This capability is crucial in a healthcare landscape where the timely delivery of equipment and supplies can have direct implications for patient safety and care quality. By optimizing inventory management and enhancing supply chain transparency, healthcare organizations can reduce spending on supplies by up to 10%, as indicated by research on supply chain management’s impact on healthcare spending.
COVR’s funding for initiatives related to the design and installation of pilot cobot systems represents another significant signal in the value chain’s evolution. These collaborative robots, or cobots, are designed to work alongside human workers, enhancing efficiency and safety in healthcare settings. The integration of cobots in patient lifting equipment manufacturing and deployment exemplifies the potential for advanced technologies to transform the value chain, offering opportunities for cost savings and improved operational performance.
The regulatory landscape, particularly the anticipated implementation of the EUDAMED database by 2027, underscores the growing importance of compliance and data transparency in the value chain. This development will necessitate that companies involved in the design, production, and distribution of patient lifting equipment adhere to stringent data reporting and safety standards, impacting how they operate and compete in the market.
As the value chain continues to evolve, companies must navigate these signals and challenges with strategic foresight and agility. The ability to adapt to regulatory changes, manage supply chain complexities, and leverage innovative technologies will be critical in shaping the future landscape of the patient lifting equipment market. These dynamics will influence market structure, pricing strategies, and competitive behavior, underscoring the intricate interplay between supply chain evolution and market outcomes.
Key Value Chain & Supply Chain Evolution Signals in Patient Lifting Equipment
In the realm of patient lifting equipment, several key signals are currently reshaping the value chain and supply chain dynamics. One of the most pressing issues is the critical shortage of drugs, which has far-reaching implications for healthcare operations, including the management of patient lifting equipment. Facilities are reportedly incurring an estimated $360 million annually in additional labor costs to mitigate the effects of these shortages on patient care. This situation reflects a broader challenge in supply chain management, where hospitals strive to maintain optimal inventory levels to prevent disruptions.
The survey conducted in 2017, revealing that 71 percent of providers were unable to offer patients their treatment of choice due to drug shortages, highlights a significant bottleneck in the healthcare supply chain. This shortage not only affects pharmaceutical availability but also underscores the potential impact on other medical equipment, including patient lifting devices. The ripple effect of such shortages necessitates robust supply chain management practices to ensure that hospitals can swiftly identify and address possible shortages or other issues related to product availability.
Furthermore, the introduction of funding initiatives like COVR for the design and installation of pilot cobot systems illustrates a strategic pivot towards innovation within the supply chain framework. These collaborative robots (cobots) are poised to enhance efficiency and safety in patient handling and lifting, thereby reshaping the operational landscape. As hospitals and healthcare facilities increasingly adopt these technologies, the demand for advanced patient lifting equipment is likely to grow, necessitating agile supply chain strategies to meet this rising demand.
Additionally, the Temporary Flaring Survey, which recorded 2,804 flaring events, although primarily related to the oil and gas industry, serves as a reminder of the importance of monitoring real-world operations to identify potential disruptions within the healthcare supply chain. The insights gained from such surveys could inform better preparedness and risk management strategies in the patient lifting equipment sector, ensuring continuity and reliability in supply chains.
Strategic Implications of Value Chain & Supply Chain Evolution in Patient Lifting Equipment
The evolution of the value chain and supply chain in the patient lifting equipment market presents several strategic implications for industry stakeholders. As the market grapples with drug shortages and the associated financial burden, companies must develop innovative solutions to enhance supply chain resilience. This involves investing in advanced technologies like inventory management systems that enable real-time tracking and forecasting of equipment needs. By doing so, healthcare providers can mitigate the risks associated with supply chain disruptions and maintain the continuity of care.
The strategic shift towards patient-centered design and human-centered approaches also demands that companies prioritize regulatory compliance and safety standards. With the European Database on Medical Devices (EUDAMED) expected to become mandatory by 2027, organizations must align their operations with these regulatory requirements. This mandates transparency in data reporting and adherence to safety standards, which could influence competitive behavior and market positioning.
Moreover, the integration of cobots and other innovative technologies into healthcare settings necessitates a reevaluation of traditional operational models. Companies like Strong Arm and GOGOA Mobility Robots, which are involved in the manufacturing of patient lifting equipment, must leverage these technologies to enhance product offerings and differentiate themselves in a competitive market. The adoption of such technologies not only enhances operational efficiency but also improves patient and caregiver safety, thereby creating a compelling value proposition for healthcare providers.
In terms of bargaining power, the emergence of advanced technologies and regulatory frameworks could shift power dynamics within the supply chain. Suppliers that offer innovative, compliant, and reliable solutions may gain greater leverage in negotiations with healthcare providers, who are increasingly prioritizing safety and compliance in their procurement decisions.
Patient Lifting Equipment Value Chain & Supply Chain Evolution Forward Outlook
Looking ahead, the patient lifting equipment market is poised to undergo significant transformations driven by regulatory changes, technological innovations, and evolving patient care needs. By 2033, the integration of advanced technologies like cobots and intelligent beds is expected to become more widespread, enhancing the efficiency and safety of patient handling processes. This trend is likely to drive demand for innovative patient lifting solutions, necessitating agile and responsive supply chain strategies to meet evolving market needs.
The mandatory implementation of EUDAMED by 2027 will further reshape the competitive landscape, compelling companies to invest in compliance and data transparency. Organizations that proactively adapt to these regulatory changes are likely to gain a competitive edge, while those that lag may face challenges in maintaining market relevance.
Moreover, as the healthcare sector continues to grapple with drug shortages and the financial implications thereof, there is an urgent need for robust supply chain management practices. By leveraging advanced inventory management systems and predictive analytics, healthcare providers can enhance their ability to anticipate and mitigate potential disruptions, ensuring the continuity of patient care.
Finally, the growing emphasis on patient-centered and human-centered design approaches will drive innovation in the patient lifting equipment market. Companies that prioritize these design philosophies in their product development processes are likely to enhance patient and caregiver experiences, thereby strengthening their market position.
In conclusion, the evolution of the value chain and supply chain in the patient lifting equipment market presents both challenges and opportunities for industry stakeholders. By navigating regulatory changes, embracing technological innovations, and prioritizing supply chain resilience, companies can strategically position themselves for success in a dynamic market landscape. The ability to adapt to these changes will be critical in shaping the future of the patient lifting equipment industry, with implications for market structure, pricing strategies, and competitive behavior.
Investment Activity
Patient Lifting Equipment Investment & Funding Dynamics Overview
The landscape of investment and funding dynamics within the patient lifting equipment market is characterized by rising investment trends and high capital intensity. These dynamics are shaped by various structural drivers, which include advancements in healthcare technology, an increasing focus on logistics infrastructure, and the growing complexity of surgical operations. These factors collectively attract significant capital flows into the market, predominantly aimed at enhancing the efficiency, safety, and technological capabilities of patient lifting solutions.
A critical factor driving investment in this sector is the necessity for healthcare facilities to adapt to evolving medical needs. As surgical operations become more complex, the reliance on specialized equipment, such as advanced patient lifting devices, becomes evident. This demand translates into increased investment by hospitals and other healthcare organizations, as they seek to improve patient care and operational efficiency. The capital intensity in this market is underscored by the significant financial commitments required to develop, manufacture, and implement these sophisticated devices.
Moreover, the strategic allocation of funds in this sector is influenced by the need to address critical operational challenges within healthcare facilities. For instance, critical drug shortages impose additional labor costs estimated at $360 million annually, prompting healthcare providers to seek innovative solutions, including investments in patient lifting technologies, to optimize resource allocation and mitigate operational disruptions.
Patient Lifting Equipment Investment & Funding Dynamics Current Scenario
In the current investment and funding scenario, the patient lifting equipment market is witnessing a robust influx of capital, driven by a combination of factors that underscore its strategic importance within the healthcare industry. The high capital intensity level reflects the substantial investments required to drive innovation and enhance the capabilities of patient lifting solutions. Despite the absence of recent mergers and acquisitions activity, the market remains vibrant, with active investors channeling substantial funds into this domain. A notable example is Target’s investment of $550 million in Shipt, a same-day gig delivery company, aimed at bolstering logistics infrastructure—a crucial component in the efficient distribution of medical devices, including patient lifting equipment.
The current funding landscape is characterized by a tiered approach to financing, with Series A, B, and C funding rounds playing pivotal roles in supporting emerging and established companies within the patient lifting equipment sector. As of 2024, the average Series A funding amount stands at $18.7 million, reflecting the initial stages of capital infusion for startups focused on innovative healthcare technologies. Series B funding typically involves amounts of $30 million or more, indicating a progression in financial support as companies scale their operations and expand their market presence. Series C funding rounds, generally ranging between $30 million and $100 million, with an average of $50 million, highlight the substantial investments required to achieve significant growth milestones and secure market leadership.
The strategic allocation of funds in this sector is further influenced by regulatory frameworks and reimbursement schemes, which play a critical role in shaping investment decisions. The Transitional Coverage (Prise en Charge Transitoire or PECT) scheme, established in 2021, provides temporary reimbursement for CE-marked medical devices, including patient lifting equipment, for up to 12 months before potential permanent inclusion in the List of Reimbursable Products and Services (LPPR). This scheme ensures a level of financial predictability and reduces the risk associated with investing in new medical device technologies, thereby encouraging further capital allocation into this market.
Additionally, the improper payment rate for patient lifts, projected at 25.4% with a projected improper payment amount of $3 million, underscores the financial challenges and risks associated with this market. However, the strategic focus on addressing these inefficiencies through targeted investments in advanced patient lifting technologies and improved operational practices presents opportunities for enhanced returns and market growth.
Key Investment & Funding Dynamics Signals in Patient Lifting Equipment
The investment and funding dynamics within the patient lifting equipment market are shaped by several key signals that reflect the strategic priorities and challenges faced by industry stakeholders. One of the primary signals is the increasing complexity of surgical operations, which necessitates substantial investments in specialized equipment and techniques. Hospitals and healthcare organizations are compelled to allocate significant resources to enhance their surgical capabilities, thereby driving demand for advanced patient lifting solutions.
In parallel, the critical drug shortages experienced by healthcare facilities, costing an estimated $360 million annually in additional labor, highlight the need for efficient resource allocation and operational optimization. This operational challenge serves as a catalyst for investments in patient lifting equipment, which can play a pivotal role in streamlining patient handling processes, reducing labor costs, and improving overall healthcare delivery.
Furthermore, the structured funding landscape, characterized by various stages of financing, provides a clear framework for capital allocation within the patient lifting equipment market. The average Series A funding amount of $18.7 million, along with Series B and C funding rounds, underscores the progressive nature of financial support available to companies in this sector. These funding mechanisms enable companies to scale their operations, invest in research and development, and bring innovative patient lifting solutions to market.
The strategic implications of these investment signals are profound, as they dictate the direction of capital flows, influence competitive dynamics, and shape the future trajectory of the patient lifting equipment market. By understanding and leveraging these signals, industry stakeholders can make informed decisions regarding investment priorities, resource allocation, and strategic partnerships, ultimately driving growth and innovation in this critical sector.
Key Investment & Funding Dynamics Signals in Patient Lifting Equipment
The patient lifting equipment market is underpinned by several key investment and funding dynamics signals that are pivotal in shaping the industry’s trajectory. Among these, the complex nature of surgical operations necessitates increased investment by healthcare organizations, driving the demand for advanced lifting solutions. This demand is bolstered by the intricate interplay between funding rounds and the strategic allocation of capital towards innovative healthcare technology and medical devices.
One of the primary signals is the escalating complexity in surgical operations, which is compelling hospitals and healthcare institutions to allocate substantial resources towards specialized equipment and techniques. This trend is a significant driver in the patient lifting equipment market, as it necessitates the adoption of advanced lifting solutions that can efficiently cater to the needs of complex surgical environments. The implication here is clear: as surgical procedures become more sophisticated, the demand for precise, reliable, and technologically advanced lifting equipment will rise, consequently attracting more investment into the sector.
Funding rounds also play a crucial role in steering capital into the patient lifting equipment market. The average Series A funding amount of $18.7 million, coupled with Series B and C rounds that range from $30 million to $100 million, highlight the substantial financial backing available for companies within this space. These funding levels enable companies to scale operations, enhance research and development efforts, and introduce cutting-edge patient lifting solutions to meet the growing market demands. The strategic allocation of these funds towards innovation and technological advancement is a testament to the market’s potential and the confidence investors have in its growth trajectory.
Additionally, the Transitional Coverage (Prise en Charge Transitoire or PECT) scheme, established in 2021, provides a temporary reimbursement framework for CE-marked medical devices, including patient lifting equipment, before their potential permanent inclusion in the List of Reimbursable Products and Services (LPPR). This temporary reimbursement mechanism is crucial as it enables manufacturers and healthcare providers to adopt new technologies with minimal financial risk, thus fostering an environment conducive to innovation and growth in the patient lifting equipment sector.
Strategic Implications of Investment & Funding Dynamics in Patient Lifting Equipment
The current investment and funding dynamics in patient lifting equipment have profound strategic implications for companies and stakeholders within the industry. As capital flows into the market, driven by the demand for advanced and reliable patient lifting solutions, companies must strategically navigate the landscape to capitalize on emerging opportunities.
One of the primary strategic implications is the necessity for companies to align their product development and innovation strategies with the evolving needs of the healthcare sector. The increased complexity of surgical operations, for instance, underscores the importance of developing lifting solutions that are not only technologically advanced but also highly adaptable to varied clinical settings. Companies that can effectively meet these needs are likely to gain a competitive edge in the market, attracting further investment and expanding their market share.
Moreover, the significant funding available through various rounds presents an opportunity for companies to invest in robust research and development initiatives. By channeling resources into R&D, companies can drive innovation, enhance product offerings, and differentiate themselves in a competitive market. This approach not only positions companies as leaders in the patient lifting equipment space but also enhances their appeal to investors seeking to back promising ventures with high growth potential.
Furthermore, the PECT scheme and similar reimbursement frameworks provide strategic leverage for companies looking to introduce new products with reduced financial risk. By taking advantage of these schemes, companies can expedite the market entry of their products, gain early adoption, and establish a foothold in the market before competitors. This strategic maneuvering is crucial for companies aiming to capture a significant share of the market and secure long-term growth.
Patient Lifting Equipment Investment & Funding Dynamics Forward Outlook
Looking ahead, the investment and funding dynamics in the patient lifting equipment market are poised for continued evolution. The rising trend in investment is likely to persist as the healthcare sector increasingly prioritizes efficiency, safety, and technological advancement. This ongoing investment will drive further innovation and development within the industry, leading to the introduction of more sophisticated and effective patient lifting solutions.
One of the key areas to watch is the continued integration of advanced technologies such as artificial intelligence and robotics into patient lifting equipment. These technologies hold the potential to revolutionize patient handling by enhancing precision, reducing the risk of injury, and improving overall patient outcomes. As companies invest in these technologies, the market for patient lifting equipment is expected to experience significant growth, attracting even more capital flows and strategic partnerships.
Additionally, the global emphasis on improving healthcare infrastructure and logistics is expected to further influence investment dynamics in the patient lifting equipment market. As healthcare systems worldwide strive to enhance their capabilities and efficiency, the demand for advanced lifting solutions that can support these goals will rise. This trend will likely lead to increased collaboration between healthcare providers, equipment manufacturers, and investors, fostering a more integrated and dynamic market ecosystem.
In summary, the forward outlook for the patient lifting equipment market is characterized by a continued influx of investment, driven by the demand for innovative solutions that can address the complexities of modern healthcare environments. Companies that can effectively navigate this landscape by aligning their strategies with market needs, leveraging funding opportunities, and embracing technological advancements are well-positioned to thrive in this evolving market. As the industry progresses, the strategic implications of these investment and funding dynamics will continue to shape the future of patient lifting equipment, driving growth, innovation, and improved patient care outcomes.
Technology & Innovation
Patient Lifting Equipment Technology & Innovation Landscape Overview
The patient lifting equipment market is undergoing significant transformation driven by technological advancements and innovation. These developments are reshaping the landscape, influencing the economics of healthcare procedures, and improving patient outcomes, throughput, and adoption rates. The integration of digital health applications, AI-driven platforms, and assistive technologies is central to this evolution. As these technologies mature, they offer new capabilities for handling patients safely and efficiently, reflecting a broader trend towards smarter and more connected healthcare environments.
NVIDIA and GOGOA are key players in this space, leveraging their expertise to push the boundaries of what is possible in patient lifting and handling. NVIDIA, known for its leadership in AI and machine learning, is contributing to the development of AI-driven platforms that streamline healthcare operations, including the collation, organization, and cross-referencing of documents. This reduces preparation time and enhances decision-making processes in patient management. GOGOA, on the other hand, focuses on active exoskeletons that support mobility and rehabilitation, offering new solutions for patient handling that are both innovative and effective.
The technological maturity stage is currently in the growth phase, indicating an increasing adoption of these technologies. This growth is facilitated by moderate patent activity levels, suggesting an active yet not overcrowded field of innovation. The high innovation intensity level further underscores the rapid pace of technological development in patient lifting equipment, with companies racing to create more advanced and user-friendly solutions.
The patient lifting equipment market is also influenced by the broader healthcare industry’s shift towards digital transformation. The implementation of electronic health records and the use of AI to analyze regulatory updates ensure that healthcare providers can deliver care that is both compliant and cutting-edge. This alignment with regulatory frameworks is crucial, as it enables faster adoption of new technologies and devices, ultimately benefiting both healthcare providers and patients.
Patient Lifting Equipment Technology & Innovation Landscape Current Scenario
Currently, the patient lifting equipment market is characterized by a growing reliance on digital health applications and AI-driven platforms. These technologies are revolutionizing the way patient handling is approached, making it more efficient, safer, and tailored to individual needs. The Digital Health Applications (Digitale Gesundheitsanwendungen, DiGA) program in Germany exemplifies this trend, allowing for accelerated approval and reimbursement of digital health applications. This program not only facilitates quicker access to innovative solutions but also encourages companies to invest in developing new technologies that align with regulatory requirements.
In addition, the complexity of surgical operations is increasing, with a greater dependence on specialized equipment and techniques. This trend necessitates substantial investment by hospitals and other healthcare organizations in advanced patient lifting solutions. These investments are driven by the need to enhance patient safety and improve operational efficiency, as well as to comply with evolving healthcare standards.
The implementation and adoption of mechanical patient lift equipment in hospitals are also influenced by organizational and cultural factors. These factors are critical for improving safety outcomes and ensuring that new technologies are effectively integrated into existing workflows. Human-centered design (HCD) approaches, which have evolved from disciplines such as human factors and human-computer interaction, play a vital role in this process. By focusing on the needs and experiences of both patients and healthcare workers, HCD facilitates the development of solutions that are not only technologically advanced but also user-friendly and practical.
Moreover, during times of medical product shortages, manufacturers and suppliers face increased pressure to meet demand. This scenario highlights the importance of efficient supply chain management and the role of technology in ensuring that patient lifting equipment is readily available when needed. AI-driven platforms and other digital tools can streamline these processes, helping manufacturers respond quickly to changes in demand and maintain a steady supply of essential equipment.
Participatory co-design methods are another important aspect of the current technological landscape. By involving patients and healthcare providers in the design process, companies can create value co-creation opportunities that enhance patient involvement and reduce costs. This approach not only leads to more effective and personalized solutions but also fosters a sense of ownership and engagement among stakeholders, which can drive adoption and long-term success.
Key Technology & Innovation Landscape Signals in Patient Lifting Equipment
As we delve deeper into the key signals shaping the patient lifting equipment market, it becomes evident that strategic investments and innovations are driving significant changes. For instance, the use of AI-driven platforms to streamline document management and regulatory compliance is a game-changer for healthcare providers. By reducing preparation time and ensuring adherence to evolving guidelines, these platforms enable more efficient and effective patient care.
Active exoskeletons, as developed by companies like GOGOA, represent another critical advancement in patient lifting technology. These devices support rehabilitation and mobility, offering patients new levels of independence and healthcare professionals more efficient ways to manage patient handling. The integration of AI and machine learning into these exoskeletons further enhances their capabilities, allowing for real-time adjustments and personalized support.
The emphasis on human-centered design and participatory co-design methods also signals a shift towards more inclusive and user-focused product development. By incorporating feedback from patients and healthcare workers, companies can create solutions that not only meet technical specifications but also address the practical challenges of patient handling in diverse healthcare settings.
Furthermore, the rising complexity of surgical operations and the corresponding need for specialized equipment highlight the importance of continuous innovation in the patient lifting equipment market. Hospitals and healthcare organizations are increasingly investing in advanced technologies to improve patient outcomes and operational efficiency, creating opportunities for companies that can deliver cutting-edge solutions.
In summary, the patient lifting equipment market is experiencing a dynamic phase of technological innovation and adoption. With key players like NVIDIA and GOGOA leading the charge, the landscape is set to evolve rapidly, offering new opportunities for improving patient care and enhancing the efficiency of healthcare operations. As these technologies mature, their impact on the market structure, demand, and competitive behavior will continue to unfold, shaping the future of patient lifting solutions.
Key Technology & Innovation Landscape Signals in Patient Lifting Equipment
The current landscape of patient lifting equipment technology is characterized by several critical signals indicative of significant shifts in the industry. These signals are not just isolated pieces of information but interconnected threads weaving a broader narrative of technological evolution. Central to these developments is the integration of digital health applications and AI-driven platforms, which promise to revolutionize patient care and enhance operational efficiency.
One of the most notable signals is the increasing complexity of surgical operations, which necessitates investments in specialized equipment and techniques. This trend underscores the need for advanced patient lifting solutions that can accommodate sophisticated surgical environments while ensuring patient safety and comfort. The demand for such equipment is likely to grow as healthcare providers strive to enhance their capabilities and address the challenges posed by more intricate procedures.
In tandem with this trend, the German Digital Health Applications (DiGA) program is accelerating the approval and reimbursement processes for digital health applications. This initiative not only facilitates quicker market entry for innovative solutions but also encourages the development of digital platforms that can seamlessly integrate with patient lifting equipment. By enabling provisional reimbursement, the DiGA program provides a financial incentive for companies to invest in the development of cutting-edge technologies, potentially leading to a surge in innovation intensity within the sector.
Moreover, the adoption of mechanical patient lift equipment is increasingly influenced by organizational and cultural factors within healthcare institutions. These factors play a crucial role in improving safety outcomes, highlighting the importance of human-centered design and participatory co-design methods. By integrating industry toolkits with patient-centered strategies, companies can enhance value co-creation, reducing costs and improving patient involvement. This approach not only aligns with the evolving needs of healthcare providers but also positions companies to better cater to the demands of a diverse patient population.
The pressure to ramp up supply during medical product shortages is another signal of the dynamic nature of the patient lifting equipment market. As manufacturers and suppliers face increased demand, they must adapt quickly to maintain supply chain resilience and meet the needs of healthcare providers. This scenario presents an opportunity for companies that can efficiently scale production and deliver reliable solutions under tight timelines.
Additionally, the WorkSafe Victoria Patient Handling Facility Unit Risk Assessment Tool (FURAT) is a critical resource that provides guidelines for the safe performance of common patient handling tasks. By offering structured methods for risk assessment, FURAT helps healthcare providers implement effective patient handling procedures, thereby reducing the risk of musculoskeletal disorders among staff and enhancing patient safety. The widespread adoption of such tools reflects a growing recognition of the importance of safety in patient lifting operations.
Strategic Implications of Technology & Innovation Landscape in Patient Lifting Equipment
The evolving technology and innovation landscape in the patient lifting equipment market presents several strategic implications for industry stakeholders. These implications are shaped by the interplay of technological advancements, regulatory frameworks, and market dynamics, each influencing the competitive landscape and shaping future growth trajectories.
One of the primary strategic implications is the need for companies to invest in research and development to stay ahead in a rapidly evolving market. As surgical operations become more complex and the demand for specialized equipment grows, companies must innovate to deliver solutions that meet the stringent requirements of modern healthcare environments. This necessitates a focus on developing AI-driven platforms and assistive technologies that can enhance the functionality and efficiency of patient lifting equipment.
Furthermore, the regulatory landscape, exemplified by programs like DiGA, offers both opportunities and challenges for companies operating in this space. While accelerated approval processes can facilitate quicker market entry, they also require companies to navigate complex regulatory requirements and ensure compliance with evolving guidelines. Companies that can effectively manage these challenges stand to benefit from first-mover advantages and capture significant market share.
The influence of organizational and cultural factors on the adoption of mechanical patient lift equipment underscores the importance of strategic partnerships and collaborations. By working closely with healthcare providers, companies can better understand the unique needs of different institutions and tailor their solutions accordingly. Such partnerships can also facilitate the co-creation of value, enhancing patient involvement and improving safety outcomes.
Supply chain resilience is another critical strategic consideration. As manufacturers face pressure to ramp up supply during product shortages, companies must ensure they have robust supply chain strategies in place to meet demand fluctuations. This requires investment in flexible manufacturing processes and the establishment of strong relationships with suppliers to ensure a steady flow of components and materials.
Patient Lifting Equipment Technology & Innovation Landscape Forward Outlook
Looking ahead, the patient lifting equipment market is poised for continued transformation, driven by advancements in technology and innovation. The integration of digital health applications and AI-driven platforms will likely play a pivotal role in shaping the future landscape, offering new capabilities for improving patient care and operational efficiency.
One of the key trends expected to shape the market is the increased adoption of smart, connected solutions that leverage AI and IoT technologies. These solutions will enable real-time monitoring and data analytics, providing healthcare providers with valuable insights into patient handling processes and facilitating continuous improvement. As hospitals and healthcare organizations increasingly prioritize data-driven decision-making, the demand for intelligent patient lifting equipment is expected to grow.
Additionally, as regulatory frameworks continue to evolve, companies will need to stay agile and adapt to changing requirements. The ability to navigate complex regulatory environments and bring innovative solutions to market quickly will be a critical differentiator for companies seeking to maintain a competitive edge.
The focus on safety and ergonomics will also remain a key consideration in the development of new patient lifting equipment. As healthcare providers strive to reduce the risk of musculoskeletal disorders among staff and enhance patient safety, there will be a growing demand for equipment that incorporates advanced safety features and ergonomic designs.
In conclusion, the patient lifting equipment market is on the cusp of significant change, driven by technological advancements and evolving market dynamics. Companies that can successfully navigate this landscape, leveraging innovation to deliver value-added solutions, are well-positioned to capitalize on emerging opportunities and achieve sustained growth. With key players like NVIDIA and GOGOA at the forefront of these developments, the future of patient lifting equipment looks promising, offering the potential to revolutionize patient care and improve healthcare outcomes.
Market Risk
Patient Lifting Equipment Risk Factors & Disruption Threats Overview
The patient lifting equipment market is subject to a variety of risk factors and disruption threats that significantly shape its market structure and dynamics. The market’s overall risk level is high, attributed to a range of structural constraints and external pressures. The necessity for patient lifting equipment in healthcare settings is driven by the need to ensure safety and reduce musculoskeletal disorders among caregivers, which are notably prevalent. However, this need is tempered by several critical risk factors that influence the market landscape.
One primary risk factor is the high incidence of musculoskeletal disorders (MSDs) among nursing assistants, as evidenced by a 2017 report indicating an incidence rate of 166.3 per 10,000 workers, which is substantially higher than the average across all industries. This underscores the pressing need for effective lifting solutions to mitigate these health risks. Despite the importance of such equipment, the market faces challenges related to improper payment practices, with a reported improper payment rate for patient lifts at 25.4% and documentation issues accounting for 91.8% of these improper payments in 2024. These financial irregularities can undermine market confidence and strain the adoption of patient lifting equipment.
Geopolitical factors also play a moderate role in this market, influencing supply chains and international trade. For instance, during periods of medical product shortages, manufacturers face increased pressure to ramp up supply, which can lead to inflated purchase orders and strain on production capabilities. The patient lifting equipment market is not immune to these geopolitical disruptions, which can impact availability and pricing.
Furthermore, substitution risks are moderate, as healthcare providers may seek alternative methods or devices for patient handling in the face of equipment shortages or financial constraints. A 2017 survey highlighted that 71% of providers could not offer a treatment of choice due to drug shortages, reflecting a broader issue of resource allocation in healthcare that could similarly affect patient lifting equipment.
Overall, the patient lifting equipment market is characterized by a delicate balance between the urgent need for effective solutions to prevent caregiver injuries and the financial and operational challenges that impede widespread adoption.
Patient Lifting Equipment Risk Factors & Disruption Threats Current Scenario
Currently, the patient lifting equipment market is navigating a complex landscape of risks and disruptions that influence its operational resilience and market behavior. The high incidence of musculoskeletal disorders among healthcare workers remains a significant driver for demand. Yet, the market’s ability to meet this demand is hampered by structural and financial inefficiencies.
The improper payment rate of 25.4% for patient lifts represents a significant financial risk, with a projected improper payment amount of $3 million. This issue is exacerbated by insufficient documentation, which accounted for 91.8% of these payments during the 2024 reporting period. Such financial mismanagement not only affects market confidence but also potentially limits the availability of funds for purchasing essential lifting equipment, thereby impacting the overall market growth.
In addition to financial challenges, the market faces operational risks related to the physical demands on caregivers. Caregivers spend approximately 25% of their time in a flexed position greater than 30 degrees when transferring patients manually, increasing their risk of injury. This highlights the urgent need for effective patient lifting solutions to mitigate these risks and improve workplace safety.
The market is also influenced by external pressures such as inflation and economic conditions. A significant number of hospitals are struggling to manage the effects of inflation and are resorting to excessive discount rates. This financial strain can lead to reduced investment in patient lifting equipment, affecting market growth and the ability to improve safety outcomes for healthcare workers.
Furthermore, the substitution risk remains moderate as healthcare providers may look for alternative solutions in the face of equipment shortages or financial constraints. The 2017 survey indicating that 71% of providers could not offer a treatment of choice due to drug shortages serves as a pertinent example of how resource scarcity can affect decision-making in healthcare, potentially impacting the adoption of patient lifting equipment.
Overall, the current scenario of the patient lifting equipment market is characterized by a high-risk environment driven by financial mismanagement, operational challenges, and external economic pressures. These factors collectively influence the market’s ability to effectively meet the growing demand for safe patient handling solutions.
Key Risk Factors & Disruption Threats Signals in Patient Lifting Equipment
The patient lifting equipment market is shaped by several key risk factors and disruption threats that influence its structural dynamics and operational viability. These signals provide insight into the challenges and opportunities within the market.
One of the most pressing risk factors is the high incidence of musculoskeletal disorders (MSDs) among healthcare workers. With a reported incidence rate of 166.3 per 10,000 workers in 2017, MSDs present a significant health risk, necessitating the adoption of effective lifting solutions. This high incidence rate is more than five times the average for all industries, underscoring the critical need for patient lifting equipment to enhance workplace safety and reduce injury rates.
Financial mismanagement further compounds the market’s challenges. The improper payment rate for patient lifts stands at 25.4%, with insufficient documentation accounting for 91.8% of these payments during the 2024 reporting period. This financial inefficiency poses a significant risk to the market, potentially limiting the availability of funds for purchasing and maintaining essential lifting equipment.
Geopolitical exposure also plays a role, albeit moderate, in shaping market dynamics. During times of medical product shortages, manufacturers and suppliers face increased pressure to meet demand, often resulting in inflated purchase orders and strained production capabilities. This can lead to supply chain disruptions and impact the pricing and availability of patient lifting equipment.
Substitution risks are present as well, as healthcare providers may consider alternative methods or devices for patient handling in the face of equipment shortages or financial constraints. The 2017 survey highlighting that 71% of providers could not offer a treatment of choice due to drug shortages illustrates the broader issue of resource allocation in healthcare, which can similarly affect the patient lifting equipment market.
In conclusion, the patient lifting equipment market is influenced by a combination of high-risk factors, including a high incidence of MSDs, financial mismanagement, and geopolitical pressures. These factors collectively impact the market’s structure, pricing power, and demand elasticity, presenting both challenges and opportunities for stakeholders within the industry.
Patient Lifting Equipment Risk Factors & Disruption Threats Current Scenario
In the current landscape, the patient lifting equipment market faces heightened operational and strategic challenges that necessitate careful navigation by industry stakeholders. The overall high risk level in this market is largely driven by structural constraints such as high incidence rates of musculoskeletal disorders (MSDs) among healthcare workers and financial inefficiencies, including improper payments. These factors significantly affect market dynamics, including pricing power and demand elasticity.
The improper payment rate for patient lifts, recorded at 25.4% with a projected improper payment amount of $3 million, underscores a critical financial vulnerability within the industry. This financial inefficiency not only affects the bottom line for providers but also influences the broader market by diverting resources away from innovation and improvement of patient safety technologies. Furthermore, insufficient documentation, which accounted for 91.8% of these improper payments, highlights systemic issues within healthcare administrative processes that could stifle market growth if not addressed.
Additionally, the high incidence rate of MSDs, recorded at 166.3 per 10,000 workers in 2017, continues to exert pressure on the healthcare industry to prioritize investments in ergonomic solutions. This rate is disproportionately higher than the average across other industries, underscoring the necessity for adoption of safer patient handling technologies. While these technologies promise to reduce physical strain on caregivers, the associated costs and implementation challenges pose significant barriers to widespread adoption.
The geopolitical exposure level of the market is considered moderate, yet it still presents notable risks, particularly as supply chain disruptions and international policy shifts can influence the availability and cost of raw materials and components necessary for manufacturing patient lifting equipment. Such disruptions can lead to increased prices and delays in product availability, impacting both supply-side stability and demand-side satisfaction.
Key Risk Factors & Disruption Threats Signals in Patient Lifting Equipment
The key risk factors and disruption threats within the patient lifting equipment market are multifaceted, arising from both internal industry dynamics and external environmental pressures. One of the most pressing internal risks is the high rate of musculoskeletal disorders among healthcare workers, as confirmed by the 2017 data showing a rate of 166.3 per 10,000 workers. This statistic not only points to a pressing health and safety issue but also emphasizes the urgency for healthcare facilities to invest in effective patient lifting solutions to mitigate these risks.
Externally, the market is influenced by broader healthcare sector challenges, such as the inability of 71% of providers to deliver a treatment of choice due to drug shortages. This signal, while primarily related to pharmaceuticals, reflects the broader resource allocation challenges within the healthcare sector, which can similarly affect the availability and prioritization of equipment like patient lifts. The strain on resources can lead to difficult decisions about which equipment to prioritize, potentially limiting investments in new technologies or upgrades.
Furthermore, the introduction of mechanical lifts and assistive devices has been shown to reduce injury rates among nursing personnel. This mitigation signal suggests a pathway to address the high prevalence of MSDs, but also indicates the necessity for continued investment and commitment to safety within healthcare environments. The adoption of such technologies is contingent upon overcoming financial and operational barriers, including the high costs associated with acquiring and implementing these solutions.
Strategic Implications of Risk Factors & Disruption Threats in Patient Lifting Equipment
The strategic implications of these risk factors and disruption threats are profound, influencing market behavior and strategic decision-making across the patient lifting equipment industry. Companies operating in this space must navigate a complex landscape characterized by high risk, regulatory demands, and competitive pressures.
For manufacturers, the imperative is to innovate and develop cost-effective, efficient patient lifting solutions that can address the high incidence of MSDs while also appealing to healthcare providers facing financial constraints. This requires a dual focus on product development and cost management, as well as strategic partnerships with healthcare facilities to foster adoption of new technologies.
Healthcare providers, on the other hand, must balance the need to invest in patient safety equipment with broader financial and resource management challenges. The high improper payment rate for patient lifts suggests that providers need to implement stronger financial oversight and documentation practices to reduce waste and improve resource allocation. Furthermore, by investing in patient lifting equipment, providers can potentially reduce the long-term costs associated with workplace injuries and improve overall operational resilience.
The geopolitical and supply chain risks also necessitate strategic planning and risk management. Companies must diversify their supply chains and explore alternative sourcing options to mitigate the impact of potential disruptions. Additionally, staying informed about international policy shifts and trade regulations will be crucial for maintaining competitive advantage and ensuring continuity of supply.
Patient Lifting Equipment Risk Factors & Disruption Threats Forward Outlook
Looking forward, the patient lifting equipment market is poised to navigate a complex array of challenges and opportunities. The high risk environment will continue to demand adaptive strategies from industry players, emphasizing innovation, financial discipline, and strategic partnerships.
As healthcare facilities increasingly recognize the importance of ergonomic solutions in reducing MSDs, there will likely be greater demand for advanced patient lifting technologies. However, this demand will be tempered by ongoing financial constraints and the need for cost-effective solutions. Providers will need to make strategic decisions about equipment investments, balancing immediate costs with long-term benefits in terms of reduced injury rates and operational efficiencies.
Manufacturers will need to focus on developing versatile, scalable solutions that can meet the diverse needs of healthcare providers. This may involve leveraging emerging technologies such as robotics and AI to enhance the functionality and efficiency of patient lifting equipment. Additionally, manufacturers will need to navigate regulatory hurdles and ensure compliance with safety standards to facilitate market entry and adoption.
The geopolitical landscape and supply chain dynamics will continue to pose challenges, requiring companies to adopt proactive risk management strategies. By building resilient supply chains and staying agile in response to international developments, companies can better position themselves to withstand external pressures and capitalize on emerging opportunities.
In conclusion, the patient lifting equipment market is characterized by high risk and significant disruption threats, driven by internal industry challenges and external environmental factors. By understanding and strategically responding to these risks, industry stakeholders can enhance their operational resilience, optimize resource allocation, and ultimately drive growth in a demanding and dynamic market landscape.
Regulatory Landscape
Patient Lifting Equipment Regulatory & Policy Environment Overview
The regulatory and policy environment surrounding patient lifting equipment is a cornerstone in shaping the market dynamics within this sector. This dimension is not merely a backdrop; rather, it serves as a pivotal force that dictates compliance, safety standards, market entry barriers, and competitive landscapes. As patient lifting equipment is essential for ensuring patient safety and enhancing healthcare delivery, the regulations and policies governing this market are designed to uphold stringent standards, ensuring that only devices meeting the highest quality and safety criteria are made available. This regulatory framework is continuously evolving, responding to technological advancements, safety concerns, and the need for harmonization across different jurisdictions.
At the heart of this regulatory landscape are several key European Union regulations, including Regulation (EU) 2017/745 on medical devices, Regulation (EU) 2017/746 on in vitro diagnostic medical devices, and the upcoming Regulation (EU) 2024/1689, which focuses on artificial intelligence. These regulations are enacted by the European Parliament and Council, underscoring the importance of a unified approach to medical device regulation across EU member states. These frameworks set the stage for compliance requirements that manufacturers must meet, encompassing everything from product design and clinical evaluation to post-market surveillance and reporting mechanisms. The emphasis is placed on ensuring that devices are not only effective but also safe for both patients and healthcare providers.
Moreover, the regulatory environment is influenced by international standards set forth by bodies such as the International Organization for Standardization (ISO). ISO standards play a crucial role in defining the technical specifications and quality management systems that manufacturers must adhere to in the design and production of patient lifting equipment. The interplay between European regulations and international standards creates a comprehensive regulatory ecosystem that governs the lifecycle of medical devices, from initial conception to market introduction and beyond.
Patient Lifting Equipment Regulatory & Policy Environment Current Scenario
The current regulatory scenario for patient lifting equipment is marked by a complex interplay of European regulations, international standards, and national policies that together shape the market. Regulation (EU) 2017/745, which governs medical devices, and Regulation (EU) 2017/746, which applies to in vitro diagnostic medical devices, provide the foundational legal framework. These regulations stipulate the requirements for conformity assessment procedures, clinical evaluations, and the roles of notified bodies in overseeing compliance.
One of the key features of the current regulatory environment is the emphasis on post-market surveillance. This is a critical mechanism designed to ensure ongoing compliance and safety of medical devices once they are in use. Manufacturers are required to establish rigorous post-market surveillance systems to monitor device performance, report adverse events, and implement corrective actions when necessary. This proactive approach not only safeguards patient safety but also enhances the credibility and reliability of patient lifting equipment in the eyes of healthcare providers and patients.
The implementation of the European Database on Medical Devices (EUDAMED) is another significant development in the current regulatory landscape. Expected to become mandatory around 2027, EUDAMED will serve as a centralized hub for information on medical devices, facilitating greater transparency and traceability. This database will enable regulatory authorities, manufacturers, and healthcare providers to access critical information on device safety, performance, and compliance, thereby streamlining regulatory oversight and decision-making processes.
Additionally, national policies such as the Transitional Coverage (Prise en Charge Transitoire or PECT) scheme in France and the Digital Health Applications (Digitale Gesundheitsanwendungen, DiGA) program in Germany are shaping the reimbursement landscape for medical devices, including patient lifting equipment. The PECT scheme provides temporary reimbursement for CE-marked medical devices for up to 12 months, offering manufacturers a pathway to market entry while awaiting permanent inclusion in the List of Reimbursable Products and Services (LPPR). Similarly, the DiGA program allows for accelerated approval and reimbursement of digital health applications, fostering innovation and adoption of new technologies.
The current scenario is further characterized by the impending enactment of Regulation (EU) 2024/1689, which lays down harmonized rules on artificial intelligence. This regulation is poised to impact the patient lifting equipment market by setting standards for AI-enabled devices, ensuring that they meet safety and performance criteria. As healthcare increasingly integrates AI technologies, this regulation will play a crucial role in defining the parameters for AI applications in patient lifting equipment, thereby influencing product development and market entry strategies.
Key Regulatory & Policy Environment Signals in Patient Lifting Equipment
In the realm of patient lifting equipment, several key regulatory and policy signals are shaping the market landscape. These signals are indicative of broader trends and shifts within the regulatory environment, providing insights into future directions and strategic considerations for manufacturers and stakeholders.
One prominent signal is the enactment of Regulation (EU) 2024/1689, which focuses on artificial intelligence. This regulation represents a significant step forward in addressing the regulatory challenges posed by AI technologies in medical devices. By establishing harmonized rules, this regulation aims to ensure that AI-enabled patient lifting equipment meets stringent safety and performance standards. This development is likely to drive innovation in the design and functionality of patient lifting devices, as manufacturers incorporate AI capabilities to enhance user experience and safety outcomes.
Another critical signal is the role of the European Parliament and Council in enacting Regulation (EU) 2017/745 on medical devices and Regulation (EU) 2017/746 on in vitro diagnostic medical devices. These regulations underscore the importance of a coherent and unified regulatory approach across EU member states. They provide a comprehensive framework for the lifecycle management of medical devices, including patient lifting equipment, by outlining requirements for clinical evaluation, conformity assessment, and post-market surveillance. This unified approach facilitates market access and ensures that devices meet consistent safety and quality standards across the European Union.
The Transitional Coverage (Prise en Charge Transitoire or PECT) scheme and the Digital Health Applications (DiGA) program are also key signals within the regulatory landscape. These national reimbursement policies highlight the intersection of regulation and market access, offering pathways for accelerated entry of innovative medical devices, including patient lifting equipment. By providing temporary and provisional reimbursement mechanisms, these programs support manufacturers in navigating the complex regulatory and reimbursement landscape, thereby fostering innovation and adoption of new technologies.
In addition to these signals, the growing emphasis on post-market surveillance and the anticipated implementation of EUDAMED reflect the regulatory focus on ongoing device performance and safety. These initiatives underscore the importance of monitoring and reporting mechanisms in maintaining the trust and confidence of healthcare providers and patients in the safety and efficacy of patient lifting equipment.
Overall, these regulatory and policy signals highlight the dynamic nature of the patient lifting equipment market, where evolving regulations and standards are shaping the competitive landscape, influencing market entry strategies, and driving technological innovation. As the regulatory environment continues to evolve, manufacturers and stakeholders must remain vigilant and adaptive to navigate the challenges and opportunities that lie ahead.
Key Regulatory & Policy Environment Signals in Patient Lifting Equipment
As the patient lifting equipment market evolves, several key regulatory and policy signals are emerging that are likely to have a profound impact on the industry. These signals form the foundation of strategic planning and market positioning for manufacturers and stakeholders. One of the most significant developments is the implementation of Regulation (EU) 2017/745, which governs medical devices within the European Union. This regulation, alongside Regulation (EU) 2017/746 for in vitro diagnostic devices, establishes a comprehensive framework that enhances device safety and effectiveness by imposing stringent requirements on manufacturers. This includes a robust conformity assessment process and the obligation for post-market surveillance, ensuring that devices continue to perform as intended after they reach the market.
The introduction of Regulation (EU) 2024/1689, focusing on harmonized rules for artificial intelligence, represents another pivotal signal. This regulation acknowledges the increasing role of AI in medical devices, setting out clear standards for its safe and reliable integration. For patient lifting equipment, which is progressively incorporating AI for enhanced functionality and user safety, this regulation is crucial. It mandates that AI components within devices must comply with specific safety and performance criteria, thereby influencing the design and development processes for manufacturers.
Another key signal is the Transitional Coverage scheme (PECT) established in France in 2021. This initiative provides temporary reimbursement for CE-marked medical devices for up to 12 months, facilitating faster market entry and enabling manufacturers to demonstrate the clinical and economic value of their devices. This temporary reimbursement scheme is particularly relevant for innovative patient lifting equipment, allowing manufacturers to gather real-world evidence that can support the case for permanent reimbursement under the List of Reimbursable Products and Services (LPPR).
In Germany, the Digital Health Applications program (DiGA) offers a parallel mechanism for accelerating the market entry of digital health technologies. By allowing for provisional reimbursement and potential extension, DiGA provides a pathway for digital components of patient lifting equipment to gain market access and reimbursement more rapidly. This is particularly pertinent for equipment that integrates digital solutions to enhance patient care and device usability.
These regulatory and policy signals collectively underscore the increasing complexity and rigor of the regulatory landscape governing patient lifting equipment. They highlight the necessity for manufacturers to invest in compliance and innovation to meet evolving standards and to leverage opportunities for market entry and reimbursement in different jurisdictions.
Strategic Implications of Regulatory & Policy Environment in Patient Lifting Equipment
The regulatory and policy environment in the patient lifting equipment sector presents both challenges and opportunities for market players. One of the primary strategic implications is the heightened barrier to entry due to stringent compliance requirements. Regulations such as the EU MDR (Regulation (EU) 2017/745) necessitate significant investments in quality assurance, clinical evaluation, and post-market surveillance. For new entrants, this could mean substantial initial costs and longer timelines to bring products to market. However, for established manufacturers who can navigate these requirements effectively, this can be a competitive advantage, creating a protective moat against new competitors.
Moreover, the regulatory emphasis on post-market surveillance and ongoing performance monitoring as highlighted by initiatives like EUDAMED, which is expected to become mandatory around 2027, necessitates continuous commitment to quality and compliance. This focus ensures that devices maintain their safety and effectiveness throughout their lifecycle, thereby enhancing brand reputation and customer trust. Manufacturers who prioritize robust post-market strategies are likely to benefit from increased market share and reduced liability risks.
The integration of AI in medical devices, governed by Regulation (EU) 2024/1689, also presents strategic opportunities for innovation. Companies that can successfully incorporate AI to enhance device functionality, usability, and safety will likely differentiate themselves in the marketplace. However, they must ensure that AI components comply with regulatory standards, which may require collaboration with AI specialists and additional investment in R&D.
Furthermore, reimbursement schemes like PECT and DiGA offer strategic pathways for market access and revenue generation. By aligning product development with the criteria set forth in these programs, manufacturers can accelerate market entry and gain early-stage revenue streams while gathering valuable evidence for permanent reimbursement. This strategic alignment can enhance a company’s financial stability and provide a competitive edge in securing market share.
In summary, the strategic implications of the regulatory and policy environment in the patient lifting equipment market revolve around compliance, innovation, and market access. Manufacturers that can effectively navigate these dimensions will likely achieve sustainable growth and competitive advantage in an increasingly regulated industry.
Patient Lifting Equipment Regulatory & Policy Environment Forward Outlook
Looking ahead to the forecast period of 2026-2033, the regulatory and policy environment for patient lifting equipment is poised to become even more complex and stringent. The anticipated mandatory implementation of EUDAMED by 2027 will likely add a new layer of transparency and data-driven oversight, encouraging manufacturers to maintain rigorous post-market surveillance practices. This development will further emphasize the importance of quality assurance and continuous improvement in device performance.
The continued integration of AI and digital health solutions within patient lifting equipment will necessitate ongoing adaptation to regulatory changes. As AI technologies evolve, regulations like Regulation (EU) 2024/1689 will likely be updated to address new safety concerns and performance standards. Manufacturers will need to stay abreast of these changes to ensure compliance and to leverage AI advancements for competitive differentiation.
Reimbursement pathways will remain a critical focus for manufacturers seeking to gain and maintain market access. Programs like PECT and DiGA may evolve to include more stringent evidence requirements, necessitating robust clinical and economic data to support reimbursement claims. Companies that invest in generating high-quality evidence and aligning their product development with reimbursement criteria will be well-positioned to secure market share and expand their customer base.
Overall, the forward outlook for the regulatory and policy environment in the patient lifting equipment market indicates a landscape that will demand greater agility, innovation, and strategic alignment from manufacturers. Companies that can navigate the complexities of this environment while capitalizing on opportunities for innovation and market access will be best positioned to thrive in the coming years. As the regulatory landscape continues to evolve, proactive engagement with regulatory bodies and investment in compliance and innovation will be crucial for sustained success in the patient lifting equipment market.
